For more than a decade, MASH — the inflammatory form of fatty liver disease — had no approved drug therapy. That changed in March 2024 when the FDA cleared Rezdiffra (resmetirom), a thyroid hormone receptor-beta agonist, as the first MASH-specific medication. Eighteen months later, on August 15, 2025, the FDA granted accelerated approval to Wegovy (semaglutide 2.4 mg) — making it the first GLP-1 receptor agonist approved for fatty liver disease, and the second drug ever cleared for MASH.
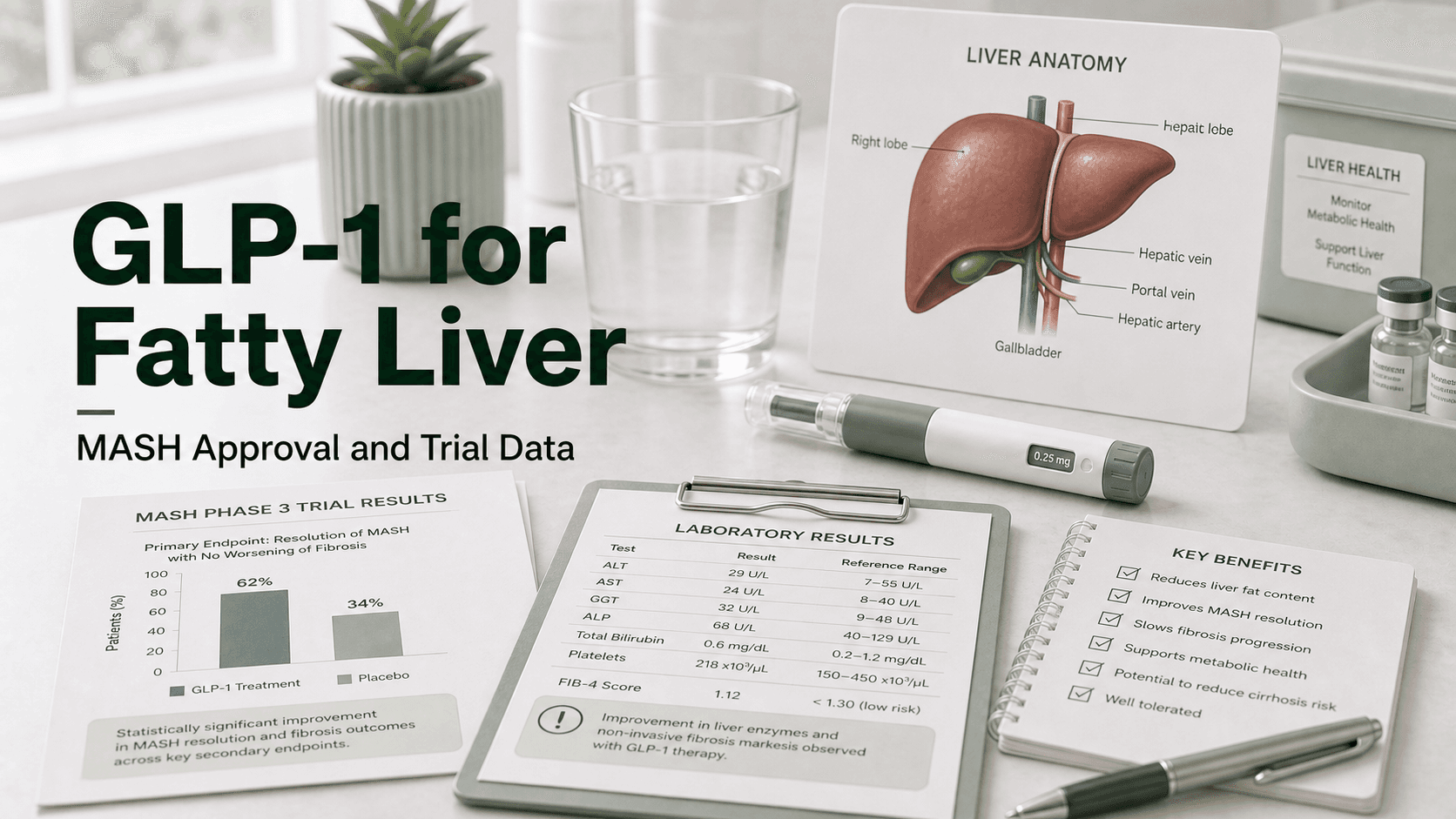
Direct answer: Wegovy (semaglutide 2.4 mg) is the only GLP-1 medication FDA approved for fatty liver disease. The label covers adults with noncirrhotic MASH and moderate-to-advanced liver fibrosis (stages F2 to F3). In the Phase 3 ESSENCE trial, 62.9% of semaglutide-treated patients had steatohepatitis resolution without worsening fibrosis at 72 weeks, versus 34.3% on placebo. A second primary endpoint showed 36.8% achieved fibrosis improvement without worsening of MASH, versus 22.4% on placebo. Tirzepatide and retatrutide are not yet approved for MASH but have produced even larger histological and imaging effects in earlier-phase trials.
Why Fatty Liver Disease Matters
Fatty liver disease sits on a spectrum. The naming has changed twice in the past few years, which is part of why patients find it confusing.
- MASLD (metabolic dysfunction-associated steatotic liver disease) — the umbrella term, replacing the older NAFLD (non-alcoholic fatty liver disease). About 38% of U.S. adults have it.
- MASH (metabolic dysfunction-associated steatohepatitis) — the inflammatory subset, replacing the older NASH (non-alcoholic steatohepatitis). Roughly 5–14% of U.S. adults have MASH; among people with type 2 diabetes, prevalence climbs to about 35%.
- Fibrosis — scarring of liver tissue, staged F0 through F4. F4 is cirrhosis.
The inflammatory and fibrotic stages — MASH with F2/F3 fibrosis — are what drive progression to cirrhosis, liver cancer, and liver-related death. That stage is exactly where Wegovy is now approved.
How the August 2025 Wegovy Approval Reads
The FDA's approval language for Wegovy specifies treatment of adults with noncirrhotic MASH and moderate to advanced liver fibrosis (consistent with stages F2 to F3). The label is structured around two histological endpoints from the ESSENCE trial: resolution of steatohepatitis without worsening of fibrosis, and improvement in fibrosis without worsening of MASH.
Key points from the label:
- Approval is accelerated — continued approval depends on confirmatory long-term outcomes data
- Used in addition to a reduced-calorie diet and increased physical activity
- Dosing is identical to Wegovy for obesity: titrate to 2.4 mg once weekly over 16 weeks
- Wegovy is not indicated for compensated cirrhosis (F4) or decompensated liver disease
ESSENCE: The Trial That Got Semaglutide Approved
ESSENCE was a Phase 3, double-blind, placebo-controlled trial that randomized 1,197 patients with biopsy-confirmed MASH and stage F2 or F3 fibrosis 2:1 to semaglutide 2.4 mg once weekly or placebo, on top of diet and exercise. The interim analysis at week 72, published in the New England Journal of Medicine in 2025, covered the first 800 patients.
Headline Histology Results at Week 72
| Endpoint | Semaglutide 2.4 mg | Placebo |
|---|---|---|
| Resolution of steatohepatitis without worsening fibrosis | 62.9% | 34.3% |
| Fibrosis improvement (≥1 stage) without worsening MASH | 36.8% | 22.4% |
| Combined resolution + fibrosis improvement | 32.7% | 16.1% |
| Mean body weight change | −10.5% | −2.0% |
Both primary endpoints hit statistical significance (P<0.001). The combined endpoint — both inflammation gone and scarring improved — is what most hepatologists consider the most clinically meaningful result, and the absolute difference there was about 17 percentage points in semaglutide's favor.
Safety in ESSENCE
By week 72, 88% of participants maintained the full 2.4 mg dose. No new safety signals emerged. GI events dominated, as expected with semaglutide:
- Nausea: 36.3% semaglutide vs 13.2% placebo
- Diarrhea: 26.9% vs 12.2%
- Constipation: 22.3% vs 8.4%
- Vomiting: 18.6% vs 5.6%
Discontinuation rates for adverse events were low and consistent with the wider semaglutide safety database.
Tirzepatide: SYNERGY-NASH Phase 2
Tirzepatide (Mounjaro/Zepbound) is not yet FDA approved for MASH, but the Phase 2 SYNERGY-NASH trial — 190 adults with biopsy-proven MASH and F2/F3 fibrosis, randomized to tirzepatide 5 mg, 10 mg, 15 mg, or placebo for 52 weeks — produced some of the strongest histology numbers reported with any therapy:
| Dose | MASH resolution without worsening fibrosis | Fibrosis improvement (≥1 stage) without worsening MASH |
|---|---|---|
| 5 mg | 51.8% | 59.1% |
| 10 mg | 62.8% | 53.3% |
| 15 mg | 73.3% | 54.2% |
| Placebo | 13.2% | 32.8% |
In responder-imputed sensitivity analyses, MASH resolution rates at the 15 mg dose reached the high-80s — which is where the often-cited "~88% resolution" figure comes from. A Phase 3 program in MASH is underway.
Retatrutide: Triple-Agonist Liver Fat Data
Retatrutide is an investigational triple agonist (GLP-1, GIP, and glucagon). The Phase 2a MASLD sub-study (Nature Medicine, 2024) used MRI-PDFF — the imaging gold standard for liver fat — and reported:
- 86% relative reduction in liver fat at the 12 mg dose at week 48
- 93% of participants on the highest dose reached normal liver fat (<5%) on MRI-PDFF, vs 0% on placebo
- Even at the 1 mg dose, 57% normalized liver fat
- Steatosis resolution in >85% of subjects with MASLD and obesity (AASLD abstract)
These are MRI-PDFF outcomes, not biopsy histology, so they do not yet prove fibrosis reversal. A dedicated MASH Phase 3 program is in planning.
Wegovy vs Rezdiffra: How the Two Approved MASH Drugs Compare
Rezdiffra (resmetirom) was the first MASH drug, approved in March 2024. Wegovy is the second. They are very different molecules, hitting MASH from different angles.
| Wegovy (semaglutide) | Rezdiffra (resmetirom) | |
|---|---|---|
| Class | GLP-1 receptor agonist | Thyroid hormone receptor-beta agonist |
| Route | Subcutaneous injection, weekly | Oral, daily |
| FDA approval | August 15, 2025 | March 14, 2024 |
| MASH resolution (vs placebo) | 62.9% vs 34.3% | ~26–30% vs 9–13% |
| Fibrosis improvement (vs placebo) | 36.8% vs 22.4% | ~24–26% vs 14% |
| Mean weight loss | ~10.5% | Modest (1–3%) |
| Other indications | Obesity, cardiovascular risk reduction | None |
| Main side effects | GI (nausea, diarrhea) | Diarrhea, nausea, pruritus |
Wegovy's histology numbers are larger than Rezdiffra's across both endpoints, and it drives substantial weight loss — which is itself a MASH disease-modifier. Rezdiffra is oral and may suit patients who cannot tolerate injectables, those who do not want to lose weight, or those who failed/were intolerant of GLP-1 therapy.
The two have not been compared head to head.
Who Qualifies for Wegovy for MASH
The label is narrower than many patients realize. To qualify under the MASH indication, an adult generally needs:
- Biopsy-confirmed MASH (or, increasingly, a strong noninvasive equivalent — FibroScan VCTE, ELF score, MRE)
- Stage F2 or F3 fibrosis (moderate to advanced, but not cirrhosis)
- Noncirrhotic liver (F4/cirrhosis is excluded)
- Inadequate response to diet and exercise alone
Patients with simple steatosis (MASLD without inflammation), F0/F1 fibrosis, or compensated cirrhosis are not covered by this indication. Many of those patients are still eligible for Wegovy under the obesity indication if they meet BMI criteria — and liver benefits would be expected secondarily.
Dosing for MASH
Dosing matches obesity:
- Start at 0.25 mg weekly for 4 weeks
- Titrate every 4 weeks: 0.5 mg → 1.0 mg → 1.7 mg → 2.4 mg weekly
- Full target dose reached around week 16
- Histology benefit in ESSENCE was assessed at week 72, suggesting at least a year of treatment before re-biopsy or reassessment
If a patient cannot tolerate 2.4 mg, the maximum tolerated dose is generally continued — though most ESSENCE responders did reach 2.4 mg.
How GLP-1s Improve Fatty Liver
The benefit comes from two converging mechanisms:
1. Weight loss. Even modest weight loss (5–10%) improves liver fat. ESSENCE patients lost ~10.5%, which is in the range where MASH resolution becomes likely. Visceral and intrahepatic fat respond preferentially to weight loss.
2. Direct metabolic effects. GLP-1 signaling reduces hepatic de novo lipogenesis, improves insulin sensitivity, lowers postprandial glucose, and reduces systemic inflammation. Some of the histology benefit in ESSENCE exceeded what weight loss alone would predict.
For tirzepatide, the additional GIP receptor activity may amplify both pathways. For retatrutide, the glucagon receptor activity directly stimulates hepatic fat oxidation — which likely explains the very large MRI-PDFF reductions.
Side Effects Specific to the MASH Population
The MASH safety profile in ESSENCE largely mirrored prior semaglutide trials. A few notes specific to fatty liver patients:
- GI events were the leading complaint, sometimes complicated by underlying NASH-related dyspepsia
- Gallbladder events (cholelithiasis, cholecystitis) occur somewhat more often with rapid weight loss — relevant in this population
- Pancreatitis risk has been studied repeatedly; the signal is small but the label still warns
- No new hepatotoxicity signal — ALT/AST trended down on semaglutide
Wegovy is contraindicated in personal or family history of medullary thyroid carcinoma and in MEN-2.
Monitoring on Therapy
Reasonable monitoring during MASH treatment:
- ALT/AST every 3 months — expect them to fall
- FibroScan or MRE every 6–12 months to track fibrosis noninvasively
- MRI-PDFF if available, especially at baseline and at 6–12 months
- Weight, BMI, waist circumference at every visit
- A1c, lipids, blood pressure — most MASH patients have multiple metabolic comorbidities
- Re-biopsy generally not required in routine practice but may be done in trials or for staging changes
- Hepatology referral for any F3 patient and for any decompensation signal
What People Get Wrong
- "Any GLP-1 treats fatty liver." Only Wegovy (semaglutide 2.4 mg) is FDA approved for MASH. Tirzepatide, retatrutide, and other GLP-1s have data but no MASH label yet.
- "I have a fatty liver on ultrasound — I qualify." The MASH indication requires inflammation and fibrosis (F2–F3), not just steatosis.
- "Wegovy reverses cirrhosis." It is approved only for noncirrhotic MASH. Compensated cirrhosis (F4) was not in the trial.
- "If my AST/ALT are normal I don't have MASH." Many MASH patients have normal liver enzymes. Imaging and elastography are how it's caught.
- "I'll just take Rezdiffra and Wegovy together." Combination MASH therapy is not labeled and not well studied. Talk to a hepatologist before stacking.
- "This replaces weight loss." It is the weight-loss tool. Diet, alcohol moderation, and exercise still matter — the label says so.
Frequently Asked Questions
What GLP-1 is approved for fatty liver? Wegovy (semaglutide 2.4 mg). FDA accelerated approval was granted on August 15, 2025 for adults with noncirrhotic MASH and stage F2 to F3 fibrosis.
How well did semaglutide work in the ESSENCE trial? At 72 weeks, 62.9% of patients on semaglutide had resolution of steatohepatitis without worsening fibrosis (vs 34.3% placebo), and 36.8% had fibrosis improvement without worsening of MASH (vs 22.4% placebo).
Is tirzepatide approved for MASH? Not yet. In the Phase 2 SYNERGY-NASH trial, tirzepatide produced MASH resolution in up to 73.3% of patients at 15 mg, and Phase 3 trials are ongoing.
What about retatrutide? Phase 2a data showed about 86% relative reduction in liver fat by MRI-PDFF at 48 weeks with the 12 mg dose. Retatrutide is investigational; MASH Phase 3 is in planning.
How does Wegovy compare to Rezdiffra? Rezdiffra (resmetirom) was the first approved MASH drug (March 2024) and works through thyroid hormone receptor-beta. Wegovy's histology numbers in ESSENCE were larger than Rezdiffra's, but they have never been compared head to head, and they may suit different patients.
Do I need a liver biopsy? Trials required biopsy. In practice, many hepatologists now use noninvasive tests — FibroScan, ELF, MRE, MRI-PDFF — to stage MASH and decide on therapy.
Can I take Wegovy if I already have cirrhosis? The MASH approval is for noncirrhotic disease only. Compensated cirrhosis from MASLD was not in ESSENCE and is not in the label.
How long until my liver improves? ESSENCE's histology endpoints were assessed at 72 weeks. Liver enzymes (ALT/AST) typically fall within the first few months. Imaging changes track weight loss, with most of the benefit appearing between months 4 and 12.
Will insurance cover Wegovy for MASH? Coverage is improving but inconsistent. Many plans now reimburse Wegovy when prescribed for MASH with documented F2/F3 fibrosis. Pre-authorization usually requires biopsy or noninvasive staging evidence.
Last reviewed: May 13, 2026
Sources
- FDA Approves Treatment for Serious Liver Disease Known as MASH — FDA
- Wegovy approved by FDA for noncirrhotic MASH with moderate to advanced liver fibrosis — Novo Nordisk press release
- Phase 3 Trial of Semaglutide in Metabolic Dysfunction–Associated Steatohepatitis — NEJM (ESSENCE)
- Semaglutide Receives FDA Approval for Treatment of MASH — Pharmacy Times
- Tirzepatide for Metabolic Dysfunction-Associated Steatohepatitis with Liver Fibrosis — NEJM (SYNERGY-NASH)
- Triple hormone receptor agonist retatrutide for MASLD: Phase 2a trial — Nature Medicine
- FDA Approves First Treatment for Patients with Liver Scarring Due to Fatty Liver Disease (Rezdiffra) — FDA
- What is the latest information on subcutaneous semaglutide for MASLD/MASH? — UIC Drug Information Group