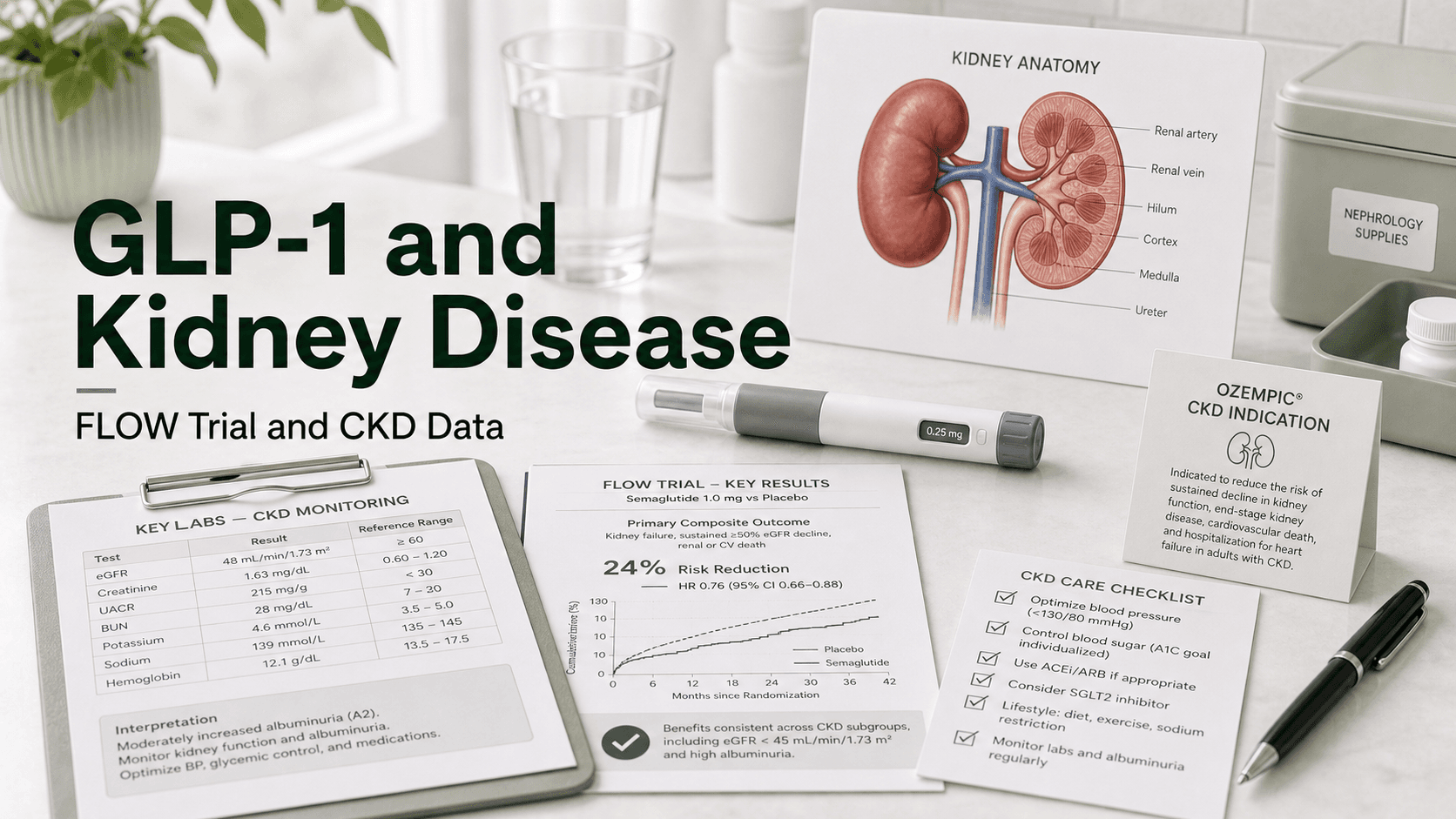
Chronic kidney disease has long had a thin shelf of disease-modifying drugs: ACE inhibitors and ARBs, then SGLT2 inhibitors, then finerenone. On January 28, 2025, the FDA added a fourth pillar — Ozempic (semaglutide) became the first and only GLP-1 receptor agonist approved to slow kidney disease progression and reduce cardiovascular death in adults with type 2 diabetes and chronic kidney disease. The pivotal FLOW trial in 3,533 patients showed a 24% reduction in major kidney events (hazard ratio 0.76), a benefit that held up across every CKD severity subgroup tested.
Direct answer: Semaglutide (Ozempic, 1 mg weekly) is FDA approved to reduce the risk of worsening kidney disease, kidney failure, and cardiovascular death in adults with type 2 diabetes and CKD. The label change came from the FLOW Phase 3 trial — 3,533 participants, median follow-up 3.4 years, primary composite of kidney failure, ≥50% eGFR decline, or kidney/CV death. Semaglutide cut that composite by 24% (HR 0.76, 95% CI 0.66–0.88, p=0.0003), slowed annual eGFR decline by 1.16 mL/min/1.73 m², reduced MACE by 18% and all-cause mortality by 20%. Crucially, the kidney protection is largely independent of weight loss — it persisted across BMI subgroups. No dose adjustment is needed for kidney function for semaglutide, tirzepatide, dulaglutide, or liraglutide; only exenatide (Byetta/Bydureon) requires renal dosing and is avoided when CrCl <50 mL/min.
Why GLP-1s Protect Kidneys
The FLOW investigators stop short of claiming a single mechanism. The leading hypotheses are:
- Reduced intraglomerular pressure — GLP-1 activation reduces sodium reabsorption in the proximal tubule, easing pressure on the glomerulus (similar in concept to SGLT2 inhibitors but via a different segment of the nephron).
- Direct anti-inflammatory effects — GLP-1 receptors are expressed in renal vasculature and tubular cells. Activation lowers oxidative stress, fibrosis signaling (TGF-β), and macrophage infiltration.
- Reduced albuminuria — UACR fell ~38% more on semaglutide than placebo, suggesting reduced glomerular barrier damage.
- Improved cardiovascular hemodynamics — Lower blood pressure, better cardiac output, and reduced atherosclerosis reduce the cardiorenal feedback loop that accelerates CKD.
- Metabolic effects — Better glycemic control (HbA1c −0.81%), weight loss (~4 kg), and BP reduction (~2 mmHg) each contribute.
The most important point for clinicians: the kidney benefit was not driven by weight loss alone. FLOW participants had a mean BMI of 32 and lost only about 4 kg on semaglutide — yet kidney outcomes improved dramatically. Subgroup analyses showed consistent benefit across BMI categories.
As trial summarizer Katherine R. Tuttle, MD put it at ADA 2024: "We know that semaglutide saves kidneys, hearts, and lives."
The FLOW Trial in Detail
FLOW was the trial that changed the GLP-1 conversation in nephrology. Run across 387 sites in 28 countries, it randomized 3,533 adults to semaglutide 1 mg weekly or placebo on top of standard of care (RAS inhibitor required, SGLT2 inhibitor optional).
Inclusion Criteria
- Type 2 diabetes with HbA1c ≤10%
- Established CKD defined by either:
- eGFR 50–75 with UACR >300 mg/g, OR
- eGFR 25–50 with UACR >100 mg/g
- On stable RAS inhibitor (ACE inhibitor or ARB) for ≥4 weeks
- Excluded: type 1 diabetes, eGFR <25, dialysis, transplant
Baseline Characteristics
- Mean age 66.6 years, 70% male
- Mean eGFR 47 mL/min/1.73 m²
- Mean BMI 32, mean HbA1c 7.8%, 61% on insulin
- Two-thirds had macroalbuminuria
- 20% had a history of heart failure
- Only 15% were on an SGLT2 inhibitor at baseline (limiting head-to-head conclusions)
Headline Results
| Endpoint | Semaglutide | Placebo | Risk reduction |
|---|---|---|---|
| Primary composite (kidney + CV death) | 5.8/100 pt-yr | 7.5/100 pt-yr | HR 0.76 (24% RRR) |
| Kidney-specific composite | — | — | HR 0.79 |
| Annual eGFR slope difference | +1.16 mL/min/1.73 m² less decline | — | p<0.001 |
| MACE | — | — | HR 0.82 (18% RRR) |
| CV death | — | — | ~29% RRR |
| All-cause mortality | — | — | HR 0.80 (20% RRR) |
| UACR change | −38% greater | — | — |
The trial was stopped early for efficacy after 741 of the planned 854 primary events accrued. That early stop, plus consistency across subgroups, is what convinced the FDA.
Safety
Fewer serious adverse events occurred in the semaglutide arm than placebo (49.6% vs 53.8%) — which sounds counterintuitive but reflects how much harm uncontrolled CKD does. Discontinuation for adverse events was 13% (semaglutide) vs 11.9% (placebo), almost entirely from GI side effects (nausea, vomiting, diarrhea). Rates of acute kidney injury and hypoglycemia were comparable.
Who Qualifies for Ozempic Under the New CKD Indication
The January 2025 label change is narrow but important. Eligible adults must have:
- Type 2 diabetes (any HbA1c — control is not required)
- Chronic kidney disease, defined by reduced eGFR and/or albuminuria
The FDA did not impose a specific eGFR cutoff in the label, but real-world prescribing tracks FLOW's enrollment — eGFR 25–75 with significant albuminuria is the sweet spot. Patients on dialysis or with eGFR <15 were not studied.
The approval does not extend to people without type 2 diabetes, even if they have CKD. It also does not extend to Wegovy (also semaglutide, higher dose) — the kidney indication is on the Ozempic label specifically.
Dose and Titration
Ozempic dosing for the CKD indication matches the diabetes label:
- Start at 0.25 mg weekly for 4 weeks (titration dose, not therapeutic)
- Increase to 0.5 mg weekly for at least 4 weeks
- Increase to 1 mg weekly — this is the FLOW-studied dose
- May increase to 2 mg weekly for additional glycemic control
The 1 mg dose is what was used in FLOW and is what the kidney/CV benefit data are anchored to. There is no requirement to push to 2 mg for the kidney indication.
Do GLP-1s Need Renal Dose Adjustments?
This is one of the most common patient and prescriber questions. The short answer is almost never.
| GLP-1 | Renal dose adjustment? |
|---|---|
| Semaglutide (Ozempic, Wegovy) | No adjustment; studied down to eGFR 15 |
| Tirzepatide (Mounjaro, Zepbound) | No adjustment; no minimum eGFR |
| Dulaglutide (Trulicity) | No adjustment |
| Liraglutide (Victoza, Saxenda) | No adjustment |
| Exenatide IR (Byetta) | Avoid if CrCl <50 mL/min |
| Exenatide ER (Bydureon) | Avoid if CrCl <50 mL/min |
| Lixisenatide | Caution if eGFR <30; avoid <15 |
Exenatide is the outlier because it is cleared renally. Most modern GLP-1 prescribing in CKD therefore uses semaglutide, dulaglutide, or tirzepatide.
The bigger practical issue is dehydration risk. GLP-1 GI side effects (vomiting, diarrhea) can drop blood pressure and reduce renal perfusion in a fragile kidney. This shows up as acute-on-chronic kidney injury in real-world data, even though FLOW did not show elevated AKI rates. Patients with eGFR <30 need slower titration, careful hydration, and a low threshold to hold a dose if GI symptoms are significant.
GLP-1 vs SGLT2 Inhibitor for Kidney Protection
This is the dominant nephrology question of 2026. Both classes are kidney-protective. Both have FDA approval for CKD with diabetes. Which to choose?
What the data show
- SGLT2 inhibitors (empagliflozin, dapagliflozin) have a longer track record in CKD, broader approvals (including non-diabetic CKD), and head-to-head observational data suggest a lower 5-year risk of CKD progression and lower rate of progression to kidney replacement therapy than GLP-1s.
- Semaglutide has the only Phase 3 dedicated kidney outcome trial for a GLP-1 (FLOW) and uniquely reduces cardiovascular death in this population.
- A 2024 JACC analysis found roughly similar kidney and cardiovascular outcomes for both classes in T2D, with a modest SGLT2i edge for 40% eGFR decline.
Practical pattern
Most nephrologists now layer these drugs rather than choose:
- Maximize RAS inhibitor (ACE inhibitor or ARB).
- Add an SGLT2 inhibitor — first-line add-on for kidney protection, works in non-diabetic CKD too.
- Add semaglutide if the patient has T2D + CKD, especially with high cardiovascular risk, albuminuria, or obesity.
- Add finerenone for residual albuminuria.
This four-pillar approach was a major theme at ASN 2024 and Kidney Week 2025.
Mechanism complementarity
The two classes act on different parts of the nephron. SGLT2 inhibitors work proximally (tubuloglomerular feedback, intraglomerular pressure). GLP-1s appear to work more through systemic metabolic, hemodynamic, and anti-inflammatory effects. The combination is therefore expected to be additive, not redundant.
Does Semaglutide Help CKD Without Diabetes?
Not yet — at least not on the label. FLOW only enrolled adults with type 2 diabetes. There is no Phase 3 outcome trial of a GLP-1 for non-diabetic CKD.
The mechanism is plausible (anti-inflammatory, anti-fibrotic, hemodynamic), and the SELECT trial showed cardiovascular benefit from semaglutide 2.4 mg in patients with obesity but without diabetes. A dedicated non-diabetic CKD trial is widely anticipated but does not yet exist. Until then, off-label use for non-diabetic CKD is not standard practice.
What About Tirzepatide and Retatrutide?
Tirzepatide (Mounjaro/Zepbound) does not have a dedicated kidney outcome trial like FLOW. The data we have are encouraging but exploratory:
- A pooled post-hoc analysis of SURPASS-1 through SURPASS-5 showed tirzepatide reduced UACR dose-dependently.
- A post-hoc analysis of SURPASS-4 showed tirzepatide nearly halved the risk of a composite kidney endpoint vs insulin glargine (eGFR decline ≥40%, kidney failure, kidney death, new macroalbuminuria).
- Tirzepatide vs dulaglutide observational data show 33% lower risk of major kidney events on tirzepatide.
That is suggestive but not approval-grade. Eli Lilly has not announced a FLOW-equivalent trial for tirzepatide. Until one happens, semaglutide is the only GLP-1 with a kidney indication.
Retatrutide (Phase 3) is too early to evaluate for kidney outcomes.
Side Effects and Monitoring in CKD Patients
The side effect profile of semaglutide in CKD mirrors the general population, with a few CKD-specific concerns:
- Nausea, vomiting, diarrhea — dehydration risk is higher in CKD; hold dose if severe.
- Acute kidney injury — rare overall but can occur via dehydration during titration.
- Hypoglycemia — risk rises if combined with insulin or sulfonylurea (61% of FLOW participants were on insulin); insulin dose often needs to be reduced.
- Gallbladder events — slightly elevated risk.
- Diabetic retinopathy — monitor in patients with pre-existing retinopathy.
- Pancreatitis — rare; standard GLP-1 monitoring applies.
Monitoring recommendations
- Baseline eGFR, UACR, electrolytes
- Recheck eGFR and UACR at 4–8 weeks after initiation, then at 3–6 months
- Watch for an early acute eGFR dip during titration — this is expected and reversible, similar to ACE inhibitor and SGLT2 initiation
- Adjust insulin and sulfonylurea doses proactively
- Continue ACE inhibitor or ARB and SGLT2 inhibitor — do not stop them when starting semaglutide
What People Get Wrong
- "All GLP-1s protect kidneys." Only semaglutide has FDA approval and a dedicated Phase 3 trial. Tirzepatide signals are suggestive, not confirmed.
- "Wegovy is approved for kidney disease." No — the CKD indication is on the Ozempic label only.
- "You need to lose a lot of weight for the kidney benefit." FLOW participants lost only ~4 kg on average. The kidney benefit is largely independent of weight loss.
- "Ozempic is for non-diabetic CKD." No — approval is restricted to T2D + CKD.
- "You have to choose between an SGLT2 inhibitor and a GLP-1." Modern guidelines support using both together in T2D + CKD.
- "My eGFR dropped after starting Ozempic — I need to stop." A modest, early drop is hemodynamic and reversible. Discuss with your nephrologist before discontinuing.
- "GLP-1s need a dose reduction if my kidneys are bad." Only exenatide does. Semaglutide, tirzepatide, dulaglutide, and liraglutide do not require renal dose adjustments.
Frequently Asked Questions
Is Ozempic approved for kidney disease? Yes — on January 28, 2025, the FDA approved Ozempic (semaglutide 1 mg or 2 mg weekly) to reduce the risk of worsening kidney disease, kidney failure, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease.
What were the FLOW trial results? FLOW randomized 3,533 patients with T2D and CKD. Semaglutide cut the primary composite of kidney failure, ≥50% eGFR decline, or kidney/CV death by 24% (HR 0.76), slowed eGFR decline by 1.16 mL/min/1.73 m² per year, cut MACE by 18%, and cut all-cause mortality by 20%.
Do I need a lower dose if my kidneys are weak? No — semaglutide does not require renal dose adjustment. The 1 mg weekly dose used in FLOW is appropriate even in moderate CKD. Only exenatide (Byetta/Bydureon) requires renal dosing.
Will Ozempic work if I don't have diabetes? The FDA approval is for T2D + CKD only. There is no Phase 3 outcome trial of any GLP-1 for non-diabetic CKD yet.
Is Ozempic better than an SGLT2 inhibitor for kidneys? Different drugs, different mechanisms. SGLT2 inhibitors may be slightly more effective at preventing eGFR decline; semaglutide uniquely cuts cardiovascular death in CKD. Most nephrologists use both together.
Does Wegovy protect kidneys too? Wegovy is semaglutide at higher doses (1.7 mg or 2.4 mg weekly). The kidney indication and FLOW data are anchored to the Ozempic label and dose. Wegovy is not labeled for CKD.
What about tirzepatide for kidney disease? Post-hoc analyses of SURPASS trials show tirzepatide reduces albuminuria and slows eGFR decline. But there is no dedicated kidney outcome trial yet, so tirzepatide is not FDA approved for CKD.
How fast does kidney function improve on semaglutide? There is usually a small early eGFR dip in the first 4–8 weeks (hemodynamic, reversible), followed by a slower long-term decline compared to placebo. The benefit accrues over years, not weeks.
Can I take semaglutide if I'm on dialysis? FLOW excluded dialysis and eGFR <25. Use in dialysis is off-label and uncommon; talk to your nephrologist.
Last reviewed: May 13, 2026
Sources
- Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes — NEJM (FLOW trial)
- FLOWing with the SemagluTIDE — NephJC trial review
- FDA Approves Semaglutide to Reduce Kidney Disease, Cardiovascular Death Risk in Adults With T2D and CKD — Pharmacy Times
- FLOW trial demonstrates kidney, cardiovascular benefits of semaglutide — ADA Meeting News
- 6 things to know about GLP-1 RAs and CKD — American Kidney Fund
- SGLT2 Inhibitors and GLP-1 RAs in Diabetic Kidney Disease — Diabetes & Metabolism Journal review
- Tirzepatide Slowed Progression of Chronic Kidney Disease — American Diabetes Association
- Tirzepatide Associated With Reduced Albuminuria — Diabetes Care (SURPASS pooled analysis)