GLP-1 Contraindications: The Complete Absolute and Relative List
The GLP-1 receptor agonist class carries an FDA boxed warning — the most serious labeling category — for thyroid C-cell tumor risk. Beyond that, a small list of conditions absolutely rules out treatment, and a longer list requires explicit risk-benefit conversation, monitoring, or specialty input. Confusing "absolute" with "relative" contraindications leads either to patients taking a drug they shouldn't, or avoiding one they would have done well on.
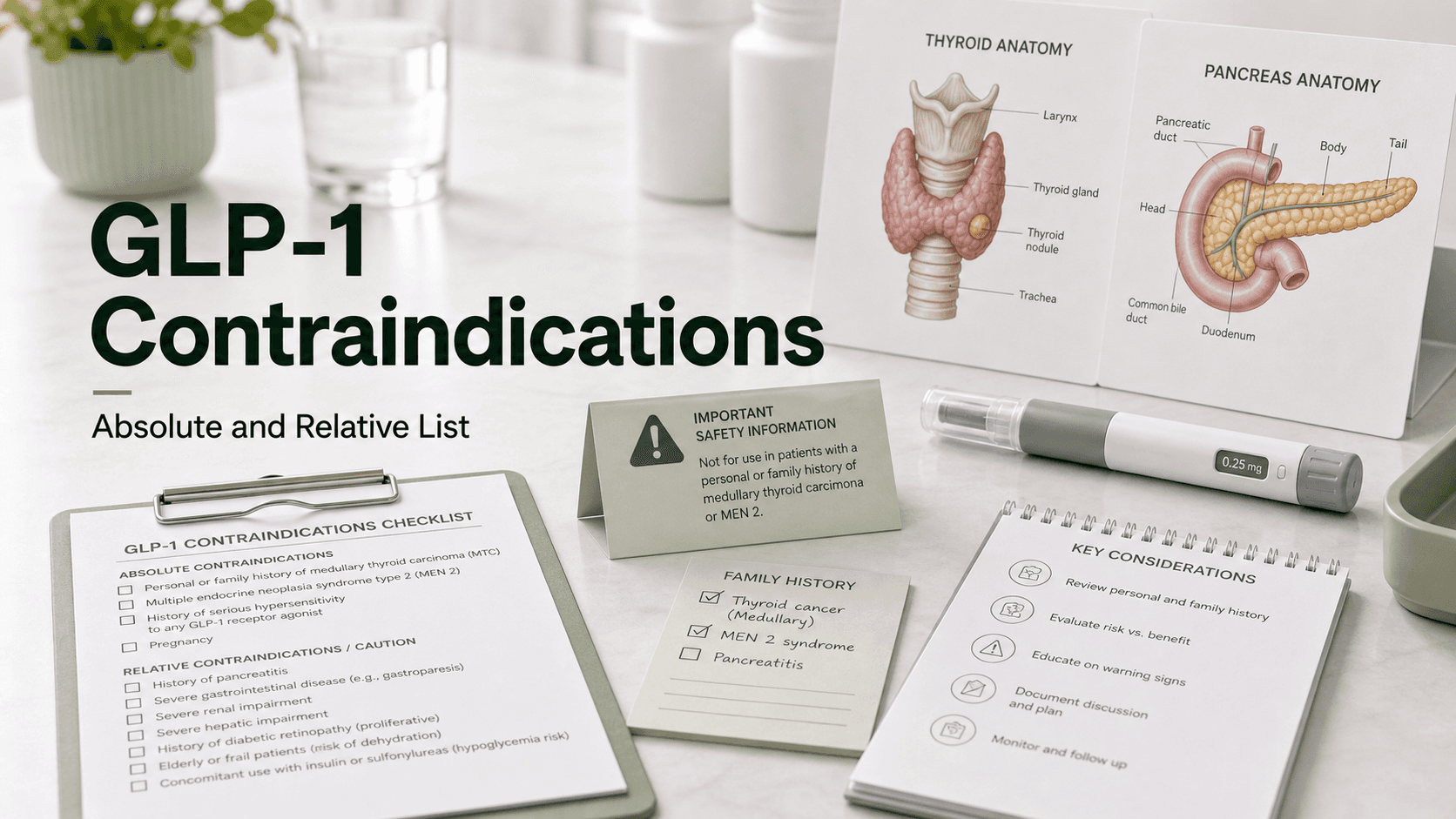
Direct answer: Absolute contraindications to all FDA-approved GLP-1 receptor agonists (Ozempic, Wegovy, Mounjaro, Zepbound, Rybelsus, Foundayo, Saxenda) are: personal or family history of medullary thyroid carcinoma (MTC), multiple endocrine neoplasia type 2 (MEN2), serious prior hypersensitivity to a GLP-1 (anaphylaxis or angioedema of the face/tongue/throat), pregnancy, and active breastfeeding. Relative contraindications — "discuss explicitly, often proceed with monitoring" — include history of pancreatitis, severe gastroparesis, active gallbladder disease, type 1 diabetes (off-label only), severe inflammatory bowel disease, active diabetic retinopathy, history of suicidal ideation, and certain bariatric surgery contexts.
Absolute Contraindications
These mean: do not start the drug.
Medullary Thyroid Carcinoma (MTC) and MEN2
The black-box warning. In rats, GLP-1 receptor agonists caused thyroid C-cell tumors. Human data has not confirmed this signal, but given MTC's severity and rarity, the FDA contraindicates GLP-1 use in:
- Personal history of medullary thyroid carcinoma
- Family history of MTC (any degree of relation)
- Multiple endocrine neoplasia type 2 (MEN2)
- C-cell hyperplasia (pre-cancerous condition)
A simple, benign thyroid nodule on its own is not a contraindication. Only the cancer or syndromic forms are.
Note: GLP-1 medications mildly increase calcitonin levels — a marker for MTC. This is one reason MTC families are excluded.
Serious Hypersensitivity Reaction
A previous anaphylaxis or angioedema (sudden swelling of the lips, face, tongue, or throat) to a GLP-1 means no GLP-1 of any kind. Cross-reactivity within the class is real, so a serious reaction to one GLP-1 disqualifies you from all of them.
Pregnancy
Animal data show fetal harm. Guidelines:
- Stop a GLP-1 at least 2 months before trying to conceive (semaglutide has a ~1-week half-life and remains active in the body for ~5 weeks after the final dose)
- Discontinue immediately if pregnancy occurs unexpectedly
- Do not initiate in anyone pregnant
Breastfeeding
Insufficient human data on transmission through breast milk. Most labels recommend avoiding use during lactation; oral Wegovy specifically is contraindicated in breastfeeding because of the SNAC absorption enhancer.
Relative Contraindications
These mean: discuss explicitly; sometimes proceed with monitoring.
History of Pancreatitis
GLP-1 agonists carry a rare but real association with acute pancreatitis. The data is mixed on whether risk meaningfully rises in patients with prior pancreatitis. Most prescribers will:
- Avoid initiating within 6–12 months of an episode
- Consider cautiously in remote, single-episode cases
- Educate patients on red-flag symptoms (severe upper abdominal pain radiating to the back, vomiting)
Rapid weight loss on a GLP-1 also increases gallstone risk — and gallstones are themselves a pancreatitis trigger.
Severe Gastroparesis
GLP-1s slow gastric emptying. In patients with significant gastroparesis, this can:
- Worsen symptoms — nausea, fullness, vomiting
- Cause severe constipation
- Raise aspiration risk during anesthesia
- Make symptoms permanent in a small subset of patients
Severe symptomatic gastroparesis is essentially absolute. Mild delayed gastric emptying is a relative contraindication that often requires a GI evaluation first.
Severe Inflammatory Bowel Disease
Crohn's or ulcerative colitis flares can be hard to distinguish from GLP-1-driven GI side effects. Active severe IBD is a relative contraindication; stable, treated IBD is usually fine.
Active Gallbladder Disease
Modest increases in gallstones and cholecystitis observed in trials, especially with rapid weight loss. Active inflammation or obstruction requires medical evaluation before treatment; stable, asymptomatic disease may still be treatable.
Active Diabetic Retinopathy
SUSTAIN-6 showed a small uptick in retinopathy progression with semaglutide, likely tied to rapid A1C reduction. Patients with active proliferative diabetic retinopathy should have eye stabilization first.
History of Suicidal Ideation or Self-Harm
FDA and EMA have been evaluating reports of suicidal thoughts in GLP-1 users since 2023. As of April 2026, no causal link has been confirmed in trial data — but:
- Patients with active or recent suicidal ideation need close monitoring
- Mental health screening before starting is increasingly common
- Family and prescriber should be informed of any worsening mood
Type 1 Diabetes
GLP-1s are not FDA-approved for T1D. Off-label use is sometimes considered as adjunct therapy, but:
- The boxed warnings still apply
- Insulin management becomes more complex (hypoglycemia risk)
- DKA risk can be elevated with significant calorie reduction in T1D
Chronic Kidney Disease
Most GLP-1s do not require dose adjustment for kidney function. Semaglutide is FDA approved to slow CKD progression in T2D. Caveats:
- Severe GI side effects can cause dehydration and acute kidney injury — monitor in patients with low baseline kidney function
- Exenatide is contraindicated in severe CKD; the rest of the class is generally OK
Severe Hepatic Impairment
Most GLP-1s are eliminated by enzymatic degradation, not the liver. Severe liver disease is not a contraindication for most patients. Practical advice: avoid in decompensated cirrhosis until specialist guidance.
Older Adults
Not a contraindication, but increased risk of lean muscle mass loss during weight loss in patients already at risk for sarcopenia. Mitigated with:
- Adequate protein (1.2–1.6 g/kg body weight per day)
- Resistance training 2–3× weekly
- Slower titration
Children Under 12
Most GLP-1s are not approved under age 12. Liraglutide (Saxenda) is approved from age 10. Semaglutide and tirzepatide are approved for adolescents (12+) in some markets. Always check the indication for the specific drug.
Bariatric Surgery History
Compatibility depends on the type:
| Surgery | Compatibility with GLP-1 |
|---|---|
| Gastric sleeve | Usually compatible; often used to maintain or accelerate loss |
| Gastric bypass | Compatible, but altered absorption affects oral formulations |
| Duodenal switch | Caution — significant motility changes |
| Adjustable gastric band | Compatible, common combination |
Talk with the bariatric surgeon, especially in the first 12 months post-op.
Conditions That Are NOT Contraindications (Despite Common Confusion)
- Simple benign thyroid nodules — only MTC and MEN2 are contraindications
- Other thyroid cancers (papillary, follicular) — not contraindicated by current labeling
- Mild GERD — often improves on GLP-1, though can worsen for some
- Gallstones in remission — caution, but not a hard contraindication
- Hypertension — improves with weight loss
- Mild-to-moderate kidney disease — usually fine; semaglutide is approved for CKD in diabetes
- Most autoimmune conditions — not contraindicated unless GI involvement is severe
- Most other diabetes medications — usually compatible (insulin and sulfonylureas need dose adjustment to avoid hypoglycemia)
Drug Interactions to Discuss
GLP-1s don't have many serious drug interactions, but several are worth specific attention:
| Class / Drug | Issue | Action |
|---|---|---|
| Insulin and sulfonylureas | Hypoglycemia risk | Usually need dose reduction |
| Oral contraceptives | Slowed gastric emptying may reduce absorption | Use barrier method for 4 weeks during tirzepatide titration |
| Warfarin | Slowed gastric emptying may alter absorption | Monitor INR during weight loss phases |
| Levothyroxine | Oral semaglutide's empty-stomach rule conflicts | Coordinate timing carefully |
| Antibiotics with absorption rate considerations | May see slowed onset | Adjust as needed |
| Digoxin | Delayed absorption | Monitor levels |
| Other GI motility drugs | Compounded slowing effects | Discuss explicitly |
Pre-Surgical Guidelines
Many surgical societies have issued GLP-1 protocols since 2023 due to aspiration risk from delayed gastric emptying during anesthesia. Common guidance:
- Pause weekly GLP-1s for 7+ days before elective surgery
- Pause daily GLP-1s for 24 hours before elective surgery
- Pre-op clear liquid diet for 24 hours if anesthesia is planned
- Coordinate with anesthesia team for any procedure with sedation
Emergency surgery on a GLP-1 patient is treated as "full stomach" for aspiration precautions.
Side Effect Rate Context
For context on what "common" looks like at therapeutic doses:
- Nausea occurs in roughly 44% of semaglutide trial participants during titration
- Effects typically improve within 2–4 weeks at stable dose
- Discontinuation due to side effects: 3–10% depending on drug and dose
What People Get Wrong About Contraindications
- "I have a thyroid nodule, so I cannot take a GLP-1." Only MTC, MEN2, or C-cell hyperplasia are contraindications. Most thyroid nodules are not relevant.
- "My kidney disease means I cannot have one." Semaglutide is actually approved to slow CKD progression in T2D.
- "One episode of pancreatitis ten years ago disqualifies me forever." Remote, single episodes are usually a discussion, not a hard no.
- "I can stay on it through pregnancy." No. Stop two months before conception.
- "My doctor said I shouldn't have surgery on GLP-1." Many can have surgery — they just need to pause the drug for 7+ days (weekly) or 24 hours (daily) and coordinate with anesthesia.
- "It conflicts with my birth control forever." Backup method is recommended only during the first 4 weeks of tirzepatide titration in most guidance.
Frequently Asked Questions
Who absolutely cannot take a GLP-1? Anyone with personal or family history of medullary thyroid carcinoma, MEN2, C-cell hyperplasia, prior serious hypersensitivity (anaphylaxis or angioedema), pregnancy, or active breastfeeding.
Can I take a GLP-1 if I had pancreatitis years ago? Possibly, with a careful discussion. Recent episodes are usually a reason to avoid; remote single episodes are case-by-case.
Is gastroparesis a contraindication? Severe symptomatic gastroparesis effectively is. Mild delayed gastric emptying is a relative contraindication that usually requires GI evaluation first.
Can I take a GLP-1 if I have kidney disease? Often yes. Semaglutide is FDA approved to slow CKD progression in T2D.
Are there contraindications specific to Mounjaro vs Wegovy? The class-wide contraindications apply to both. Tirzepatide has the additional 4-week oral contraceptive precaution during titration. Specific package inserts include drug-specific cautions.
What about surgery? Most surgical societies recommend pausing weekly GLP-1s for 7+ days before elective surgery, daily GLP-1s for 24 hours, to reduce aspiration risk from delayed gastric emptying.
How long after stopping a GLP-1 is it safe to get pregnant? At least 2 months. Semaglutide's long half-life means it stays active in the body for ~5 weeks after the final dose.
Last reviewed: May 13, 2026
Sources
- GLP-1 Safety: Side Effects, Drug Interactions, and Who Shouldn't Take Them — Medical Foundation of NC
- Who Shouldn't Take GLP-1 Medications — Potere Health MD
- GLP-1 Contraindications Reference for Prescribers — Trimi
- GLP-1 Contraindications Safety Checklist — Peptide Dossier
- GLP-1 Agonists — Cleveland Clinic